Claim: Molnupiravir works by randomising the virus's genetic code
This insane drug has received approval in Singapore and elsewhere.
Molnupiravir: the perfect treatment for causing a new pandemic.
Molnupiravir, Merck’s anti-viral drug that has recently been approved for use in Singapore, and the UK. This is quite simply insane: it’s straight out of a Michael Crichton novel.
You see, Molnupiravir works by introducing errors into the genetic code of the virus. Mutations, in other words; the very thing we do not want.
Generally Molnupiravir would disable the virus. The problem is, sometimes, very occasionally, it might create a viable mutation that is worse than the original, and when you’re talking about this operating in one single person, that’s very many mutations being created, thousands, millions; and when you’re talking about it happening in a whole population, if just one of these mutations is more efficient than the original virus at being transmitted and/or more virulent, and then you have the perfect process for creating a worse virus than the original.
This is the chemical formula for Cytidine, which is very important in viral replication:
This is the chemical formula for Molnupiravir:
As you can see, these two formulas are very similar.
Molnupiravir disturbs viral replication by disturbing RNA polymerase, the enzyme critical for viral replication. Molnupiravir effectively inserts itself into the RNA copying process of the virus, thereby creating new mutations of the virus.
Associate professor of bioengineering and neurobiology at Stanford University Michael Z. Lin says that the worst case scenario is that
it could generate repeated cycles of new variants, with people desperately taking the drug to fight the new variants it spawns, creating a vicious positive feedback loop while causing more suffering and deaths.
About the FDA fact sheet he says that the
the fact sheet recognizes that “changes in the spike protein occurred at positions targeted by monoclonal antibodies and vaccines.” Bafflingly, however, it adds, “The clinical and public health significance of these changes are unknown.”
But Michael Z. Lin says:
The significance of changes to spike protein positions by antibodies and vaccines is very well known: These changes are what allowed each variant of concern — from alpha to beta to delta to omicron — to evade immunity from previous infection, vaccines or monoclonal antibody treatment.
The FDA understands the risks of creating new variants because in their letter to MERCK indicating the conditions of authorisation they require MERCK to monitor for new variants of the virus caused by the drug:
Merck will submit the complete viral shedding results and full genome SARS-CoV-2 nucleotide sequencing results from the full randomized population in study MK-4482-002 Part 2. Viral sequencing analyses should include all Baseline and End-of-Treatment (Day 5) samples with sufficient RNA levels for analysis, as well as all Post-Treatment samples with viral RNA levels ≥100,000 copies/mL. Cell culture infectivity assessments should be conducted for any clinical specimens in which amino acid changes were detected in the SARS-CoV-2 spike protein.
William Haseltine, a prominent virologist, gave a similar warning in a series of articles in Forbes, and referred to a series of mouse studies testing Molnupiravir against two highly pathogenic mouse viruses, MERS-CoV and the mouse hepatitis virus.
He says the virus was found to be still able to replicate after many mutations, and that outside of the lab, being filtered through millions of people, such a drug could be an unmitigated disaster:
Critically, the researchers found that the viruses could survive and replicate to high titers despite such large numbers of mutations in every gene and protein. The viruses tested did show a slight replication disadvantage — though they still replicated to the same high titers, they did so slightly less rapidly compared to the original non-mutated viruses. However, outside of the lab, as the drug is given to millions of people with active infections, this disadvantage may quickly disappear as we would likely provide a prime selection environment to improve the fitness of the virus…. We are potentially headed towards a world class disaster.
Molnupiravir (Lagevrio) is not only a potential cause of a new pandemic, it is able to cause mutations in Mammalian cells as well.
Haseltine points to this study, which finds that Molnupiravir is able to cause mutations in the cells of mammal hosts of the virus the drug is supposed to be treating.
However, the risks for the host may not be zero and these risks need to be carefully evaluated in undertaking therapies that are by their nature designed to change the coding capacity of a virus’ genetic material. Evaluating the utility of this drug should be done in those likely to receive the greatest benefit with monitoring provided to assess potential long-term genotoxic side effects.
This may be why the FDA fact sheet in particular has laughable warnings against using Molnupiravir (brand name: Lagevri) for male “sexually active individuals with partners of childbearing potential” - my comment in italics:
While the risk is regarded as low (?how do they know this?) …studies to fully assess the potential for Lagevrio to affect offspring of treated males have not been completed.
and furthermore while doctors should:
…advise sexually active individuals with partners of childbearing potential to use a reliable method of contraception correctly and consistently during treatment and for at least 3 months after the last dose of Lagevrio.
because
The risk beyond three months after the last dose of Lagevrio is unknown.
So let’s just get this straight: the risk is unknown before three months and after three months!
Oh, look, they haven’t even done the rat studies yet:
A planned study of male testicular germ cells from transgenic rats will assess the potential for molnupiravir to affect offspring of treated males.
(The other thing that’s scary about this is they have transgenic rats that create male human testicular germ cells….!!!!)
The warnings for females are just as scary:
Pregnancy Testing Prior to initiating treatment with Lagevrio, assess whether an individual of childbearing potential is pregnant or not, if clinically indicated [see Warnings and Precautions (5.1)]. Contraception Females Advise individuals of childbearing potential to use a reliable method of contraception correctly and consistently, as applicable for the duration of treatment and for 4 days after the last dose of Lagevrio
Interestingly this section is not as large, possibly because the risks are known. In the warnings and precautions:
WARNINGS AND PRECAUTIONS Embryo-Fetal Toxicity: Lagevrio is not recommended for use during pregnancy. (5.1, 8.1, 8.3)
and
Based on findings from animal reproduction studies, Lagevrio may cause fetal harm when administered to pregnant individuals. If Lagevrio is used during pregnancy, prescribing healthcare providers must communicate to the patient the known and potential benefits and the potential risks of Lagevrio use during pregnancy, as outlined in the “Fact Sheet for Patients and Caregivers”
They are scarily inexact in describing the levels of embroyofetal lethality etc:
In an animal reproduction study, oral administration of molnupiravir to pregnant rats during the period of organogenesis resulted in embryofetal lethality and teratogenicity at 8 times the human NHC (N4-hydroxycytidine) exposures at the recommended human dose (RHD) and reduced fetal growth at ≥ 3 times the human NHC exposure at the RHD. Oral administration of molnupiravir to pregnant rabbits during the period of organogenesis resulted in reduced fetal body weights at 18 times the human NHC exposure at the RHD (see Data)
Some of the reported side effects in rat studies sound particularly horrifying. Molnupiravir interferes in bone and cartilage growth:
5.3 Bone and Cartilage Toxicity Lagevrio is not authorized for use in patients less than 18 years of age because it may affect bone and cartilage growth. Bone and cartilage toxicity was observed in rats after repeated dosing.
Probably the most horrifying statement is right at the beginning of the warnings section:
WARNINGS AND PRECAUTIONS There are limited clinical data available for Lagevrio. Serious and unexpected adverse events may occur that have not been previously reported with Lagevrio use.
Yes, taking Molnupiravir (Lagevrio) might well be like giving your three year old child a blowtorch and trusting him to fix the computer in your car engine with it.
Problems with the clinical trial
The approval of Molnupiravir in the USA, Australia, Singapore, UK, and elsewhere, is highly problematic, particularly in view of repeated concerns expressed in letters to the BMJ about the quality of the clinical trials, which were truncated early for reasons that are not clear.
Addendum: FDA lies in their approval
There is no adequate, approved, and available alternative to the emergency use of Lagevrio for the treatment of mild-to-moderate COVID-19 in adults as further described in the Scope of Authorization (section II).6
What about Ivermectin plus zinc? Oh, I guess it’s a cheap generic drug and it won’t result in giant kickbacks for the big players.
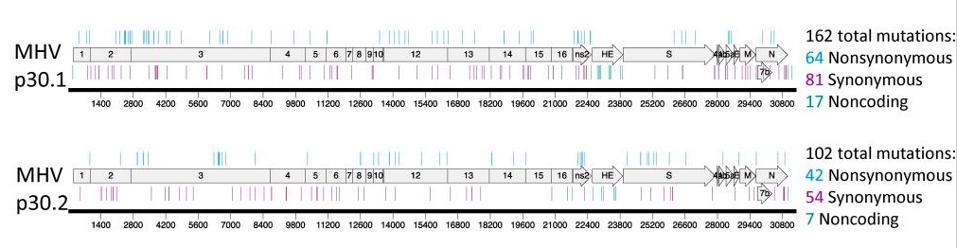
Update - new study confirms that Molnupiravir creates viable viral mutations
A new pre-print study in medRxiv examines the claim that one of the two new drugs used to treat Covid, Molnupiravir, cause viral mutations that could spread and become new Covid variants. They prove that this is true.
In a double blind trial with 9 patients, they gave Molnupiravir to 5 and another drug that does not cause genetic mutations in the virus to 4. They found that the 5 patients who had received Molnupiravir had viable Covid mutations that could be spread in the community when they went home, because all 9 patients still tested positive for Covid at the end of their hospital stay.
I actually examined this claim in earlier this year (2022) in May and will add this article to the fact check.
This is the study:
Antiviral treatments lead to the rapid accrual of hundreds of SARS-CoV-2 mutations in immunocompromised patients Nicholas M Fountain-Jones, Robert Vanhaeften, Jan Williamson, Janelle Maskell, I-Ly Chua, Michael Charleston, Louise Cooley medRxiv 2022.12.21.22283811; doi: https://doi.org/10.1101/2022.12.21.22283811
Like all preprint studies on medRxiv, the study has a large blue disclaimer pointing out that it is a preprint and should not be used to guide clinical practice.
Here is the abstract.
Abstract
The antiviral Molnupiravir (Lageviro) is widely used across the world to treat SARS-CoV-2 infection. Molnupiravir reduces viral replication by inducing mutations throughout the genome, yet in patients that do not clear the infection, the longer-term impact of the drug on virus evolution is unclear. Here, we used a case-control approach to monitor SARS-CoV-2 genomes through time in nine immunocompromised -patients with five treated with Molnupiravir. Within days of treatment, we detected a large number of low-frequency mutations in patients and that these new mutations could persist and, in some cases, were fixed in the virus population. All patients treated with the drug accrued new mutations in the spike protein of the virus, including non-synonymous mutations that altered the amino acid sequence. Our study demonstrates that this commonly used antiviral can ‘supercharge’ viral evolution in immunocompromised patients, potentially generating new variants and prolonging the pandemic.
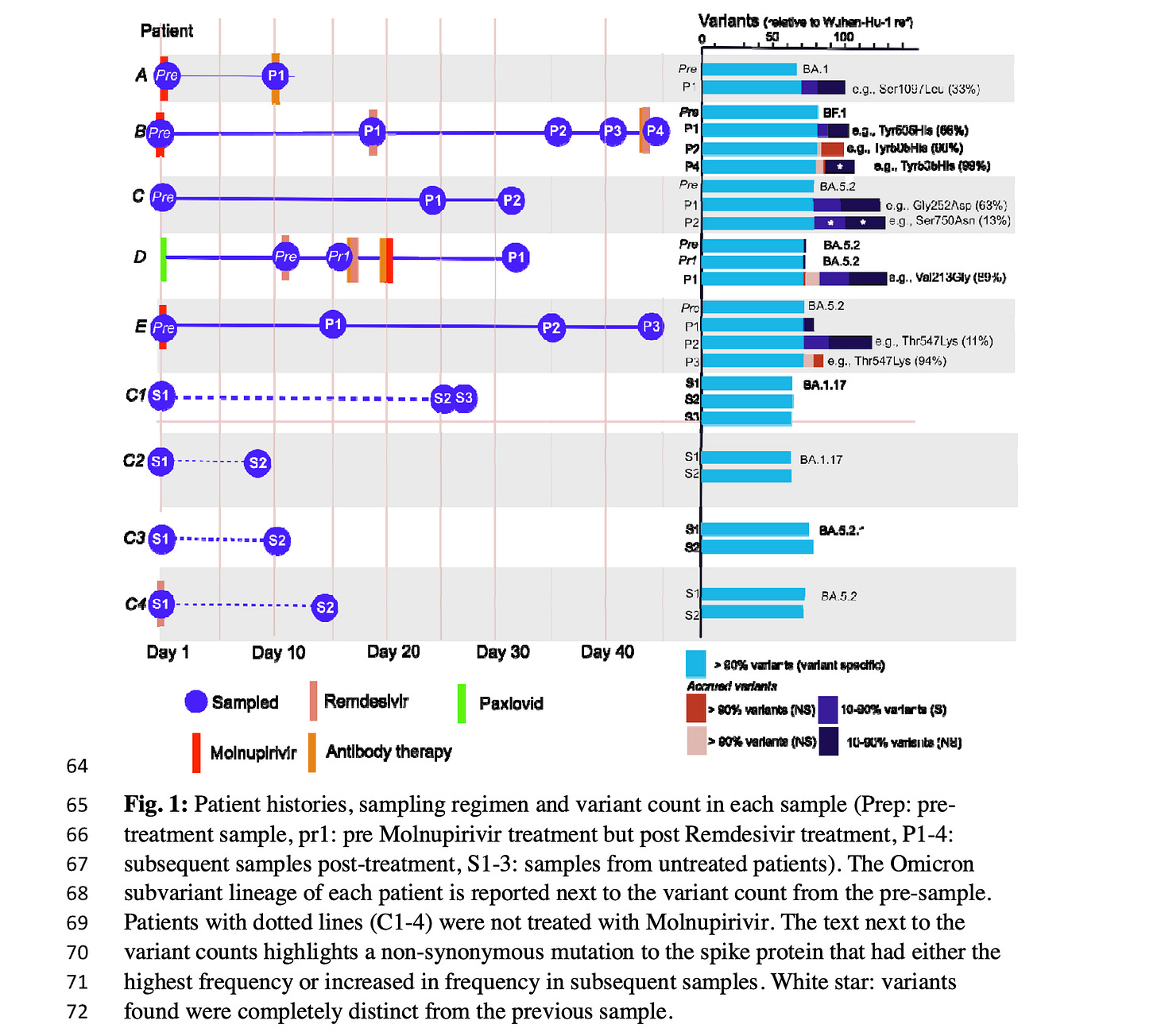
This graph shows the new variants produced in each of the Molnupiravir patients A,B,C, D and E (to the right, under “Variants”; this is not seen in the non-Molnupiravir patients C1-C4.
Their conclusion is quite damning:
These data highlight the risk of treating immunocompromised patients with error generating antivirals such as Molnupiravir. All of the individuals in our study remained persistently PCR positive post-treatment, although active monitoring for clearance was not undertaken by the institution. It is possible they were infectious in the hospital and in their communities, and onward transmission of these highly divergent viruses is likely. This commonly used class of antivirals has the capability to supercharge SARS-CoV- 2 evolution, and uncontrolled use may generate new variants with a transmission advantage that prolongs the pandemic and makes other therapeutics less effective.
CHANGELOG
Mon 16 May 5:36pm Fixed the quote re Merck monitoring for new variants.