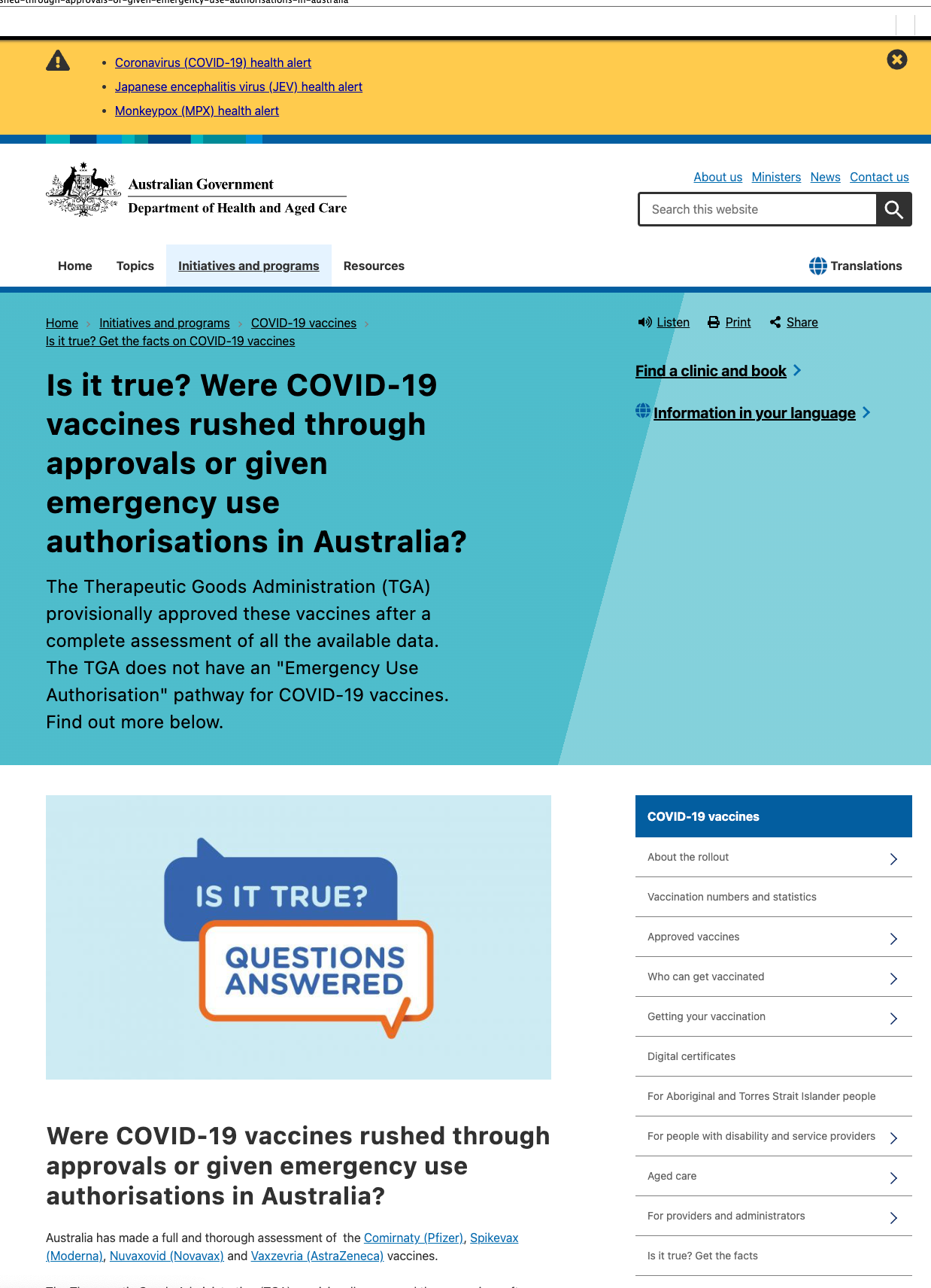
I will leave it to the reader to decide why the TGA got rid of this webpage, which claimed that the approvals were not rushed.
Here is how the webpage appeared a year ago:
Here is the page as it appears now:
Is this because of the February 2023 FOI request document release?
I’m sure it doesn’t have anything to do with the recent release of the only decent sized Pfizer BNT162b2 approval document they could find in response to an FOI request. In this document the data doesn’t match the text, a document that clearly looks rushed because it still had track changes enabled when they made the pdf version.
mRNA/LNP didn't stay at the injection site for the rats either, it went to the heart and ovaries
Looking at the FOI TGA document 6, the Nonclinical Evaluation Report BNT162b2 [mRNA] COVID-19 vaccine (COMIRNATY™) Submission No: PM-2020-05461-1-2 Sponsor: Pfizer Australia Pty Ltd January 2021. https://web.archive.org/web/20230204175226/https://www.tga.gov.au/sites/default/files/foi-2389-06.pdf
Their claim that Australia doesn’t have “Emergency Use Authorization” on that old webpage is untrue anyhow, because the TGA has “Provisional Approval” instead, which to all extents and purposes amounts to the same thing, at least insofar as I can tell.





Thanks, there's nothing really to say, just to wonder how long is it going to take to get somewhere from here.